Microscopy
Aug 13, 2014 07:19:03 #
Mike Adams
Loc: Connecticut
I know this forum is for macrophotography,, but I guess many readers will also be interested in photo microscopy. If this is not an appropriate place, please let me know.
I am using stacking software with my microscope pictures and the result is not nearly as effective as I had hoped. In many cases a single picture looks far better than the final stacked image. I think it is because, with a basically transparent object, the stack 'clutters up' the final image. Has anyone tried this, and if so, got results you are happy with.
ps I am comparing the pictures to those obtained with a confocal microscope that flattens the final stack. This may be unfair sine the confocal is using ultraviolet lasers as the light source and can achieve far better spatioal resolution.
I am using stacking software with my microscope pictures and the result is not nearly as effective as I had hoped. In many cases a single picture looks far better than the final stacked image. I think it is because, with a basically transparent object, the stack 'clutters up' the final image. Has anyone tried this, and if so, got results you are happy with.
ps I am comparing the pictures to those obtained with a confocal microscope that flattens the final stack. This may be unfair sine the confocal is using ultraviolet lasers as the light source and can achieve far better spatioal resolution.
Aug 13, 2014 07:34:53 #
Confocal Microscopes.... a whole nother world !!
http://www.physics.emory.edu/faculty/weeks//confocal/
http://www.physics.emory.edu/faculty/weeks//confocal/
Aug 13, 2014 12:34:41 #
We have had posted microscopy images in the past; we have an occasional (rare) poster who has posted images that qualify. I'd love to see some of them. I do some stacking-- but not 10x and beyond.
Aug 13, 2014 14:37:32 #
Mike Adams
Loc: Connecticut
This is a ciliate I shot earlier today. Something odd has happened to the color transferring to my desktop, but the level of resolution is correct. The length of the cell is about 100 microns
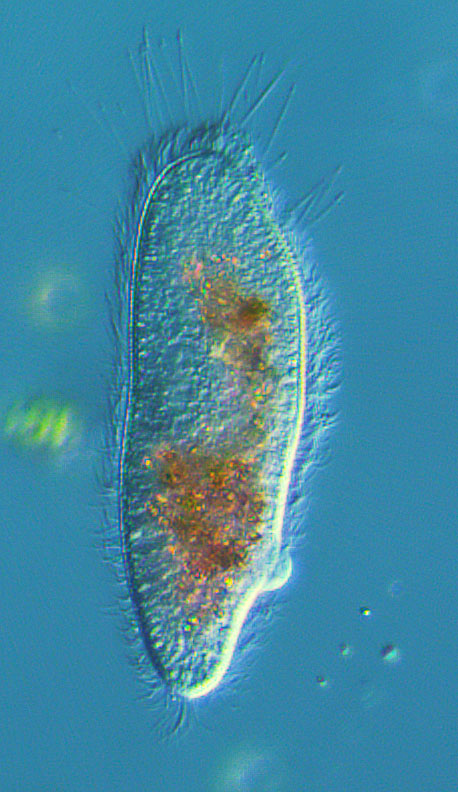
Aug 13, 2014 19:30:00 #
Mike Adams wrote:
1 micron = 0.001 millimeters (http://www.unitconversion.org/length/microns-to-millimeters-conversion.html )The length of the cell is about 100 microns
100-microns = 0.1 millimeters
We welcome your photo-micrography. Please post more.
Aug 13, 2014 22:39:26 #
Hi Mike - Welcome to the macro forum. My first thought is you have a vibration issue. The higher the magnification the more prone to vibration the captured image becomes. Not sure how much the image is cropped but it looks like you are in the 400X range. I've never shot anything at this magnification. I have shot images at 40X and vibration was an issue until I got a marble anti vibration table.
Tell us a bit about your setup and the measures you have taken to eliminate vibration.
Tell us a bit about your setup and the measures you have taken to eliminate vibration.
Aug 14, 2014 08:17:57 #
Mike Adams
Loc: Connecticut
A-PeeR wrote:
A good point. WHile the entire complex is on a floating foundation there may be internal vibration. The scope is sitting on a rubber pad, that should help, but I am not in a position to go to an air table. The shutter release is controlled electronically and the exposure time may be the culprit, about 1/10 of a second. Since I can't boost the lighting any further, I might try upping the ISO to get faster speeds.My first thought is you have a vibration issue. The higher the magnification the more prone to vibration the captured image becomes. Not sure how much the image is cropped but it looks like you are in the 400X range. I've never shot anything at this magnification. I have shot images at 40X and vibration was an issue until I got a marble anti vibration table.
Tell us a bit about your setup and the measures you have taken to eliminate vibration.
Tell us a bit about your setup and the measures you have taken to eliminate vibration.
Aug 14, 2014 08:59:09 #
Mike Adams wrote:
What make and model of microscope is this? What camera are you using and how is it attached to the scope? I am currently in the process of setting up a Lietz Diavert Trinocular for pond water observation and photographing specimens. The light that came with the unit is woefully inadequate for photography. I bought a used lamp housing off eBay and I am fashioning it to hold a Canon 430-EX II flash. Idea being I can view the specimen with the normal lamp to pick starting and ending frames points. Replace the replace the lamp housing with the "flash" housing to shoot a stack with speedlite illumination. Still working on the housing adapatation (can never seem to find enough time), perhaps you can boost illumination with a similar adaptation?A good point. WHile the entire complex is on a floating foundation there may be internal vibration. The scope is sitting on a rubber pad, that should help, but I am not in a position to go to an air table. The shutter release is controlled electronically and the exposure time may be the culprit, about 1/10 of a second. Since I can't boost the lighting any further, I might try upping the ISO to get faster speeds.
Aug 14, 2014 12:36:17 #
Mike Adams
Loc: Connecticut
I am using an Olympus B40 with DIC optics. The camera may be the weak point, it is a SPOT 3MB that mounts onto the video tube and uses its own software package (does not allow Raw images). I would use my Nikon D5300 but that model cannot alter the aperture without a lens attached. I doubt if I will upgrade since the D5300 was really bought for macro work.
Aug 15, 2014 00:31:54 #
A BH40 or a BX40, with the former you should be able to pick up a used light housing on the cheap and modify for flash. DIC optics, that explains 3D effect of the photo. I wouldn't swap out the camera just yet. You should be able to get really good photos with those objectives and a properly aligned system. See this article for the basics of setting up DIC:
http://www.olympusmicro.com/primer/techniques/dic/dicconfiguration.html
As for setting the aperture on your 5300, you don't need to. Aperture is set by the microscope objective. Doesn't really matter what the camera thinks aperture is. The camera is coupled to a trinocular tube or photo port without a lens.
http://www.olympusmicro.com/primer/techniques/dic/dicconfiguration.html
As for setting the aperture on your 5300, you don't need to. Aperture is set by the microscope objective. Doesn't really matter what the camera thinks aperture is. The camera is coupled to a trinocular tube or photo port without a lens.
Aug 20, 2014 08:03:48 #
Mike Adams
Loc: Connecticut
Its a BX. I am not really in a position to make changes to it anyway; the department lets me use it, but on sufferance. This is all new to me, I started just a couple of months ago and have virtually no background in microscopy, so I have a steep learning curve. Thanks for the link, very informative.
Yes, I can adjust the speed on the D5300, but cannot see the results of what I am doing. So, I shoot blind at different settings and look at the results.
As an alternative to DIC, I hope to try dark field illumination, as well as some vital stains. The sludge at bottom of our pond is providing a range of interesting subjects.
Yes, I can adjust the speed on the D5300, but cannot see the results of what I am doing. So, I shoot blind at different settings and look at the results.
As an alternative to DIC, I hope to try dark field illumination, as well as some vital stains. The sludge at bottom of our pond is providing a range of interesting subjects.
If you want to reply, then register here. Registration is free and your account is created instantly, so you can post right away.



